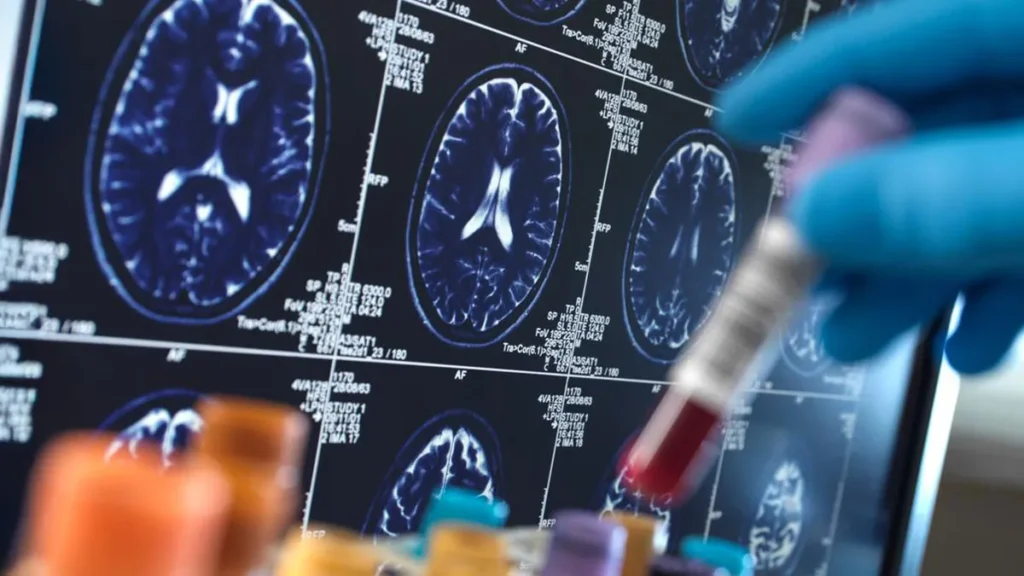
A new technique called Transcranial Radio Frequency Stimulation is capable of neuromodulation, show NYU Langone’s neuroscience and radiology researchers.
The two diseases cause damage in some of the same ways.
A new study, led by researchers at NYU Langone Health, addresses the moments when we first recognize a blurry object, a primal ability that enabled our ancestors to avoid threats.
Researchers at NYU Langone Health and scientists at Fermilab are exploring whether quantum computing can help bring about long envisioned quantitative MRI. Step one: teaching qubits (and qudits) to do the math.
Imaging researchers at NYU Langone have used deep learning to turn noise against itself in order to improve low-field MRI. They’re after something much bigger than sharper images.
Goodbye, FMR9
A fond look at the legacy and the final moments of an MRI scanner that played a special role in building NYU Langone’s imaging research program.
NYU Langone welcomes an MRI research system for conducting in vivo investigations of the brain’s microenvironment with unprecedented sensitivity.
Computed tomography (CT) scans of the chest, abdomen, and spine, taken originally to detect problems such as kidney stones or growths on the lungs, can be repurposed through artificial intelligence (AI) to catch signs of bone loss, a new study shows.
Imaging scientists at NYU Langone have created an AI model that assesses MRI data during the exam to inform the remainder of the imaging session.
Lavanya Umapathy, postdoctoral fellow who develops representation learning models for medical imaging, talks about improving prostate-cancer screening and using artificial intelligence to approach “the person behind the images.”